Laryngeal Dystonia (LD) - also called spasmodic dysphonia - is a voice disorder that leads to strained or choked speech. Current therapeutic options for treating LD are very limited. LD does not respond to conventional speech therapy and is treated primarily with Botulinum toxin injections to provide temporary symptom relief. There is no cure for LD.
Vibro-tactile stimulation of the larynx – A novel, non-invasive behavioral therapy to treat the voice symptoms of LD
Vibro-tactile stimulation (VTS) is a non-invasive neuromodulation technique that our laboratory developed for people with LD. In a first study supported by the National Institutes of Health, our team documented that a one-time 30-minute application of VTS can result in measurable improvements in the voice quality of people with LD. The scientific results of this study have been published and can be viewed here.
A clinical trial study to assess the longitudinal effects of VTS
In a second research study also funded by the National Institutes of Health, we investigated systematically the possible longer-term benefits of this approach for improving the voice symptoms of people with LD. Study participants administered VTS at home by themselves for up to 8 weeks. In addition, researchers assessed their voice quality and monitored the corresponding neurophysiological changes in the brain using electroencephalography in the laboratory at the beginning, in the middle and at the end of the VTS in-home training. The findings of this study showed that up to 57% of participants showed a 30% improvement or more in an objective voice measure (Cepstral Peak Power) or a reduction in perceived voice effort. The results inform patients and clinicians about the possible impact of this therapeutic approach. It promotes the development of wearable VTS devices that would enlarge the available therapeutic arsenal for treating voice symptoms in LD.
This study has been registered at ClinicalTrials.gov ID: NCT03746509
Note: This clinical trial has stopped recruiting new participants.
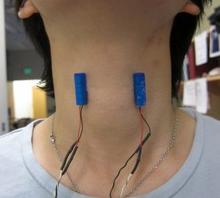

NEW - A usability study to determine in-home use of laryngeal VTS
The study aims to get insights in how people with laryngeal dystonia can use our vibration device at home over a 2-month period. It does not require a lab visit, but participants will follow a specific study protocol and record their user experience. For more detailed information, visit the clinicaltrials.gov website here.
This study has been registered at ClinicalTrials.gov ID: NCT06111027.
If you are interested in participating in a usability study using the vibration device, sign up through this link: https://z.umn.edu/VibrationDevice
Contact us
If you want to stay updated on the latest progress, please sign up through this link: https://z.umn.edu/SDSignUp
Team members
Other members of the interdisciplinary research team include Dr. Peter Watson, a voice disorder specialist, and Dr. Yang Zhang, an expert in the analysis of cortical activity during speech. Both are faculty in the U of M Department of Speech, Language and Hearing Sciences. Dr. George Goding from Otolaryngology represents the clinical partner in the team. He is an expert in LD and treats these patients regularly in the U of M Lion’s Voice Clinic. Dr. Divya Bhaskaran, Dr. Naveen Elangovan, and other members from HSCL complement the team.


Contact information
Dr. Jürgen Konczak, Ph.D. is the principal investigator of these studies. You can contact him at [email protected].
Watch webinar from Dysphonia International